Table Of Content
- Overview of drug development in the United States of America
- The last component: maximizing benefits from the information collected from a valid trial
- ALND can be safely omitted for patients with sentinel-node macrometastases
- The argument framework is a flexible approach to evidence in healthcare
- Trial Stages and Designs
- Competing events

Clinical trial design is an important aspect of interventional trials that serves to optimize, ergonomise and economize the clinical trial conduct. The purpose of the clinical trial is assessment of efficacy, safety, or risk benefit ratio. A well-conducted study with a good design based on a robust hypothesis evolved from clinical practice goes a long way in facilitating the implementation of the best tenets of evidence-based practice. A robust well-powered trial adds to the meta-analyzable evidence base and contributes huge quanta to our knowledge of dermatological practice. This article sets out to describe the various trial designs and modifications and attempts to delineate the pros and cons of each design and attempts to provide illustrative samples for the same where possible. Blinding is a fundamental tool in clinical trial design and a powerful method for preventing and reducing bias.
Overview of drug development in the United States of America
Flare Therapeutics Presents FX-909 Phase 1 Dose Escalation and Expansion Clinical Trial Design at the 2024 ASCO ... - PR Newswire
Flare Therapeutics Presents FX-909 Phase 1 Dose Escalation and Expansion Clinical Trial Design at the 2024 ASCO ....
Posted: Fri, 26 Jan 2024 08:00:00 GMT [source]
Different viral variants have also emerged during the pandemic, which may also influence outcomes, as have effective vaccines. For these reasons, concurrent controls are critical as a means to reduce temporal bias, though this feature is not common to all platform trials during the COVID-19 pandemic. In our view, this lesson should be an enduring one that lingers even after the pandemic has drawn to an end. Firstly, significance of the composite does not necessarily imply significance of the components nor does significance of the components necessarily imply significance of the composite. For example one intervention could be better on one component but worse on another and thus result in a non-significant composite.
The last component: maximizing benefits from the information collected from a valid trial
When the primary objective of a trial is to evaluate non-inferiority, an ITT analysis skews the data towards potentially showing non-inferiority. Because of this it is typical in non-inferiority trials to use an as-treated analysis as the primary analysis [39, 40]. The goal of this paper is to provide an initial guidance to investigators through the design process of a clinical trial.
ALND can be safely omitted for patients with sentinel-node macrometastases
The trial is working to automate data collection from electronic medical records in order to ease the burden of conducting time-sensitive research when resources may be overstretched. I-SPY COVID is also unique in its biomarker development initiative, which incorporates the collection and study of biospecimens to investigate the biologic heterogeneity of severe COVID-19 that may influence outcomes and/or treatment effects9. This is the ideal type of study to run, assuming there is enough preliminary data to effectively evaluate risks and benefits (and that the benefit/risk assessment is not based on wishful thinking or assumptions). Depending on the robustness of previous data, phase II, phase III or pivotal trials with clinically relevant outcomes can be run. For example, in Bolognini et al., 5 days of tDCS (a technique thought to be safe (48) that has been studied in neuropathic pain) led to a significant and sustained reduction of pain levels in PLP patients, along with an improved ability to move their limbs (10).
Cohort studies are study designs that compare two groups, such as the subjects with exposure/risk factor to the subjects without exposure/risk factor, for differences in incidence of outcome/disease. Most often, cohort study designs are used to study outcome(s) from a single exposure/risk factor. Thus, cohort studies can also be hypothesis testing studies and can infer and interpret a causal relationship between an exposure and a proposed outcome, but cannot establish it (Figure 4). One way to see the importance of this problem is by analyzing the attrition rate from early to late phases. Attrition means the drop in the number of drugs that make it to market compared to those studied in preclinical and clinical trials.
The argument framework is a flexible approach to evidence in healthcare
Because of the increased probability of a false-positive conclusion discussed above all secondary objectives and analyses in a clinical trial should interpreted as non-definitive or hypothesis generating. When many “secondary” analyses are provided after the primary endpoint of a trial is not met, the results of any “significant” findings should be viewed with strong skepticism or outright ignored. Regression analyses are an important element of randomized trials analyses even when the primary analysis is not based on regression models.
There can be many logistic and ethical concerns in clinical trials where neither a placebo, nor a sham control can be applied. Blinding is especially important in studies where subjective response are considered as outcomes. This is because certain responses can be modified based on the knowledge of the experiment group that they are in. For example, a group allocated in the non‐intervention group may not feel better as they are not getting the treatment, or an investigator may pay more attention to the group receiving treatment, and thereby potentially affecting the final results.
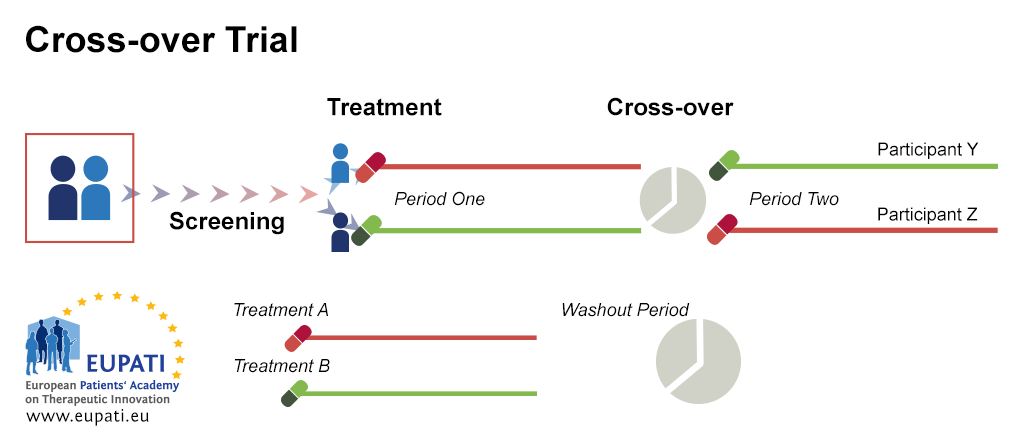
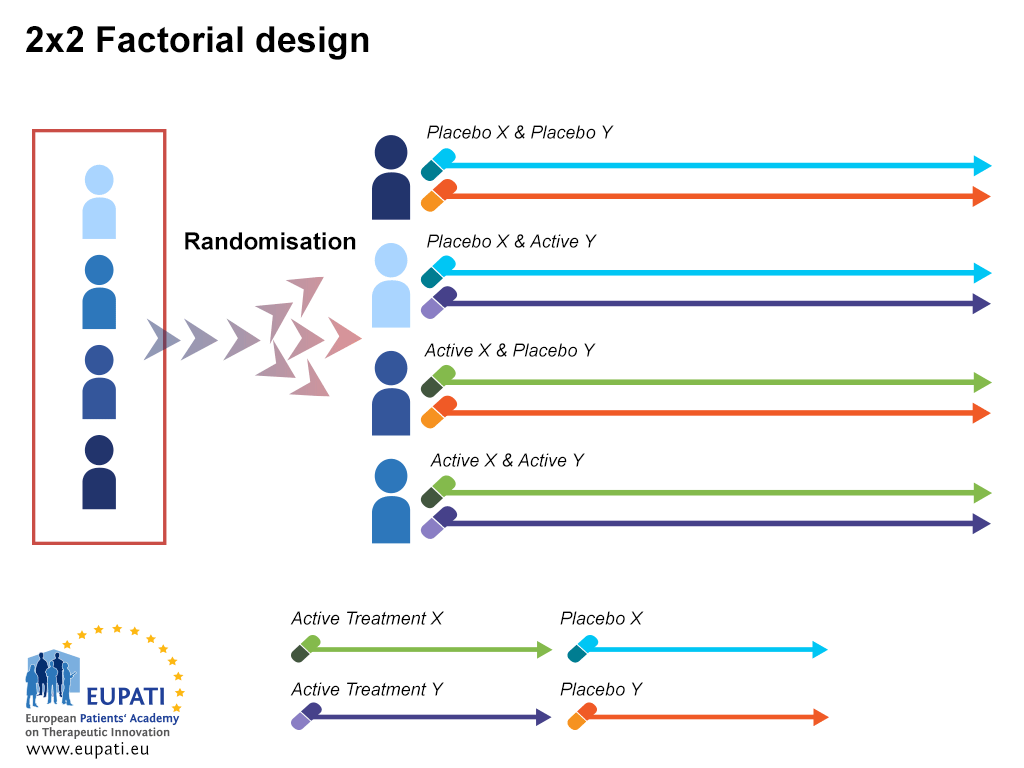
Trial Stages and Designs
However, when historical data are very reliable, well documented and other disease and treatment conditions have not changed since the historical trial was conducted, then they can be considered. Historical controls have become common in device trials when placebo-controls are not a viable option. Historical controls can be helpful in interpreting the results from trials for which placebo controls are not ethical (e.g., oncology trials).
Trials participants must be treated with the utmost respect, and that begins with careful attention to informed consent. In this module, you’ll learn about the personnel, documents, terminology, and practices that go into planning and implementing ethically sound informed consent procedures within your trial. To the Editor — It is difficult to forget the chaos, anxiety and heightened urgency of early 2020, when COVID-19 became a global pandemic. Infections around the world were skyrocketing, while clinicians faced tremendous uncertainty over how to treat this devastating new infection. Hundreds of different therapeutic approaches were proposed, many of which had a relatively weak link to the pathophysiology of COVID-19, our understanding of which was rapidly evolving. Without data to guide clinicians, tens of thousands of patients received a wide range of untested therapies.
Can Patient-Led Clinical Trial Design Save Neurology Research - Clinical Leader
Can Patient-Led Clinical Trial Design Save Neurology Research.
Posted: Wed, 17 Apr 2024 07:00:00 GMT [source]
Significance level refers to the p-value threshold for concluding statistically significant results; it also corresponds to the type I error rate (the chance of concluding an effect when in fact none exists). In general, larger sample sizes are needed to detect a smaller effect size, achieve greater power, and/or reduce the type I error rate. In addition to these factors, sample size calculations for trials should anticipate loss to follow-up and withdrawals, patient non-compliance to treatment, and protocol violations and ineligibility.
In this way they aimed to help target patient subpopulations that might benefit from specific interventions, rather than group different PLP subsets to receive a therapy that might only works on a small number of them (44). Such a mechanism-based approach may help direct clinical trials toward positive outcomes. For this reason, the NIH proposed a mechanistic data model to design psychiatry trials – the RDoC (Research Domain Criteria) framework (45). Phase III trials provide additional information about effectiveness, efficacy and safety, giving a clearer picture of the intervention’s risk-benefit ratio with a potential to change clinical practice (21).
The Company also announced acceptance of its HB-200 study abstract as an oral presentation at the American Society of Clinical Oncology (ASCO) 2024 Annual Meeting in the head and neck cancer session to be held on June 4, 2024. The presentation will include data for approximately 40 head and neck cancer patients treated with HB-200 in combination with pembrolizumab. The Company anticipates the first patient will be enrolled in the fourth quarter of 2024. Further, it is important to specify parameters for all stopping rules for stopping the trial early. In a Frequentist design, stopping rules are defined in terms of boundaries for safety, efficacy and futility; see Chapter 8 in Ellenberg, Fleming, and DeMets (2002). In a Bayesian design, stopping rules are typically defined in terms of posterior probabilities or predictive probabilities; see Saville et al. (2014).
The scale of the primary endpoint impacts the analyses, trial power, and thus costs. Cohort studies can be classified as prospective and retrospective.7 Prospective cohort studies follow subjects from presence of risk factors/exposure to development of disease/outcome. This could take up to years before development of disease/outcome, and therefore is time consuming and expensive.

No comments:
Post a Comment